Create of Clinical Trial Material
"CCTM"
Clinical trial material creation by EP-Link’s specialists
EP-Link’s specialists with CRC experience assigned for the CCTM service get an understanding of the protocol from an early stage and can also provide insight from a CRC perspective to provide optimal service. The CCTM service ensures that the quality of clinical trial materials is improved and costs are reduced.
Benefits of Using the CCTM Service
-
Reduction in workload of Clinical Research Monitors (CRAs)
EP-Link’s support for the creation of clinical trial materials can reduce CRA workload and costs.
-
Quality consistency across clinical trial materials
The quality of the trial materials can be standardized as they are created by EP-Link’s specialists. EP-Link has the ability to create clinical trial materials in a wide range of therapeutic areas, including oncology, which is one of EP-Link’s strengths, and can respond quickly to the needs of a global trial team.
Operation Structure for CCTM Service
EP-Link has established a dedicated unit for creation of clinical trial materials at its headquarters. EP-Link selects the most suitable team leader for each project and organizes a team. Along with the team leader, experienced CRCs (CCTM staff), DMs (data and image organizers), and quality review staff are assigned to quickly provide the clinical trial materials that meet the sponsor’s requirement. The team leader serves as the point of contact to meet the sponsor's needs.

CTTM Service Includes:
- To-do List
- Worksheets (WS)
- List of prohibited concomitant medications and restricted medications
- Pocket-size protocol
- Window table
- Configuration list of a case file
- Title page and spine
- Others (Specimen processing instructions, etc.)
- * EP-Link provides the service to create the individual or complete set of these materials quickly and flexibly to meet the specific needs of each sponsor.
Experience of CTTM Service
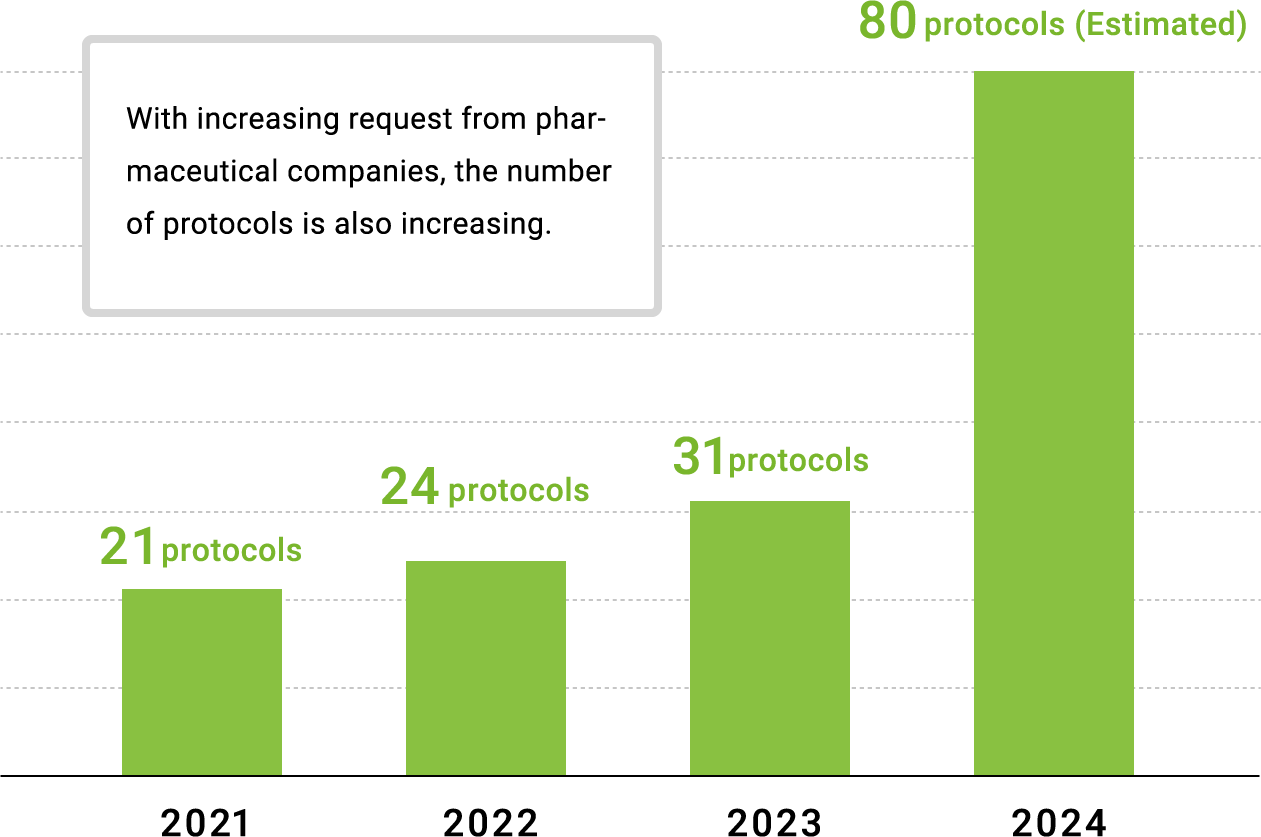
Client’s comment: Company A (Clinical Development personnel)
-

Client’s comment: Company A (Clinical Development personnel)
CCTM Service was very helpful because they collated and confirmed details, and even pointed out discrepancies in protocols and some documents. As a result, in addition to preparing the study materials, CCTM Service helped us to make some minor corrections. I was also very impressed with their QC capabilities.
-

Client’s comment: Company B (Clinical Development personnel)
Thank you for creating well-organized worksheets. Overall, the deliverables were logically structured and of excellent quality.
-

Client’s comment: Company C (Clinical Development personnel)
Thank you for providing such level of work in a short period of time. We are really impressed.


.png)
@2x.png)
@2x.png)
.png)
@2x.png)
@2x.png)
.png)